Rep:Mod:slippynipps2
Module 2 Inorganic Computational Lab
Nicholas Browning
Introduction
Computational chemistry provides insight into the structure and bonding of complexes This is of great use in inorganic chemistry as often metal complexes are hazardous to handle in the lab and so computational chemistry provides a much safer and more enjoyable route than conducting the experiment in a wet lab. Computational chemistry also allows us to accurately calculate the Molecular Orbitals of any given molecule, given enough time. This is a huge resource for all chemists as molecular orbital interactions underpin all chemistry and understanding their function in a visual and calculative sense is vital. This project conducts the following experiments:
- Analysing the structural, vibrational and Molecular Orbital properties of two molecules of the type XL3.
- Rationalise the structural and vibrational properties of a large metal complex by simplifying the calculation through ligand substitution.
- Rationalise and compare the observed stabilities of three dimeric lewis acids through the use of structural, vibrational and Molecular Orbital discussions.
Small Molecules XL3
Borane BH3
Geometry Optimization
Borane was drawn in GaussView and an geometry optimization was then run using the B3LYP method with a 3-21G basis set. The results are shown in Table 1.
| Bond length | 1.19Å |
| Bond Angle | 120.0o |
| Energy | -26.46226 a.u. (-69,480 kj/mol) |
The output file (Available Here) provides the following information about this optimisation:
- The calculation type is FOPT.
- The file type is .log.
- The calculation method is RB3LYP
- The basis set is 3-21G.
- The dipole moment is 0.00 Debye.
- The gradient is 1.07 x 10-6 a.u., which is less than 0.001 a.u. meaning the optimization succesfully reached a stationary point.
- The forces had converged, also showing a sucesful optimisation.
- A study of the frequency analysis shows that the energy is at a minima and all the low frequency vibrations are positive.
Frequency Analysis
BH3 is a non-linear molecule and hence is expected to have 6 (3N-6) vibrational modes. These modes were calculated using the following criteria:
# freq b3lyp/lanl2dz geom=connectivity
The results are tabulated in Table 2.
One would therefore expect 6 peaks to appear on the experimental IR spectrum of BH3, however the mode at 2598.42 cm-1 is IR inactive as the vibration itself is symmetrical and leaves no change in dipole moment. There are also two pairs of degenerate vibrations at 1203.64cm-1 and 2737.44cm-1. The IR spectrum is shown below.
Molecular Orbitals of Borane
One of the powerful aspects of Molecular Orbital Theory is that it can be used in both a qualatative and quantatative sense.First, the qualative representation LCAO approach will be applied to BH3 to generate the MO diagram. Then a calculation will be run to determine the precise geometry and energies of the orbitals determined by the qualatative approach. This then allows us to compare the two approaches and comment upon strengths and weaknesses of each approach.
Qualitative Approach
The MO diagram of BH3was produced using LCAO theory and was drawn in ChemDraw. The diagram is shown below:

Quantitative Approach
The MO's of BH3 were calculated using the following parameters:
# b3lyp/3-21g pop=(nbo,full) geom=connectivity
The resulting Orbitals and their energies are shown in Table 3. The output file is available [here].
| MO | 1a1' | 2a1' | 1e' | 1e' | 1a2" | 2e' | 2e' | 3a1' |
| LCAO representation |  |
 |
 |
 |
 |
 |
 |

|
| GaussView picture |  |
 |
 |
 |
 |
 |
 |

|
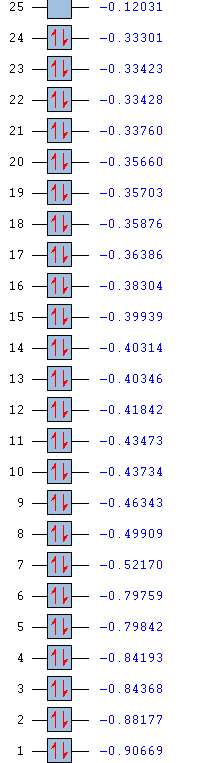
| Energy | -6.73023 | -0.51778 | -0.35689 | -0.35689 | -0.07455 | 0.18879 | 0.18879 | 0.19236 |
There is a good correlation between the qualitative LCAO's and the calculated MOs. In order to get an accurate representation of the MO, each LCAO must be superimposed with a specific contribution factor that determines how much that LCAO contributes to the overall MO. Clearly in order to gain this contribution factor for large molecules a computational calculation must be run hence somewhat defeating the purpose of the qualitative approach. Although the qualitative approach gives a good approximation of the MO's for small molecules, it is increasingly less accurate as the molecule gets larger in size and geometric complexity.
Natural Bond Orbital

The Natural Bond Orbital was calculated, and gives a representation of the charge distribution within the molecule. The molecule is neutral, and hence the total charge of the molecule must add up to 0, as shown in the log file. The log diagram shows that all hydrogen's are electrostatically equivalent, which is expected given the symmetry of the molecule. Similarly it also shows that the boron is electropositive comparatively to the hydrogen's due to it's lewis acid nature.
Furthermore, the log file gives information about the hybridisation state of the atoms and their subsequent effect on the molecules MO's. It states that the three bonding orbitals of the boron atom have a 33.33% contribution from the s-orbital and a 66.66% contribution from the p-orbitals, which shows that the boron is sp2 hybridised. These boron hybrid orbitals contribute 4.5% of the B-H bonds and the hydrogen orbitals contribute the remaining 55.5%. This indicates that the bonding MO's are distorted somewhat toward the hydrogen's, which is in agreement with the charge distribution present within the molecule.
Thallium Bromide TlBr3
Geometry Optimization
The geometry optimisation was calculated using the B3LYP method with the LANL2DZ basis set. The resulting output files can be found here(.chk) and here(.log). The symmetry of thallium (III) bromide is very tightly restricted to the D3h point group and hence the optimisation produced an output with very clear D3h symmetry, having a bond angle of 120.0o. The output had converged with a gradient of 6.08 x 10-11 a.u which is less than 0.001 a.u suggesting a successful calculation.
The bond length of 2.65Å is in good correlation to the literature value of 2.5153Å [1] which shows the accuracy of this calculation is good on small molecules. The differences between the calculated and literature values is most likely due to the inaccuracies inherent within the basis set, and could further be improved by using a more detailed one.
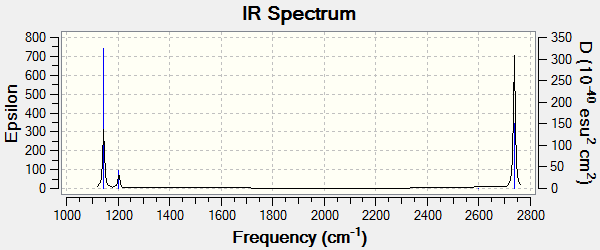
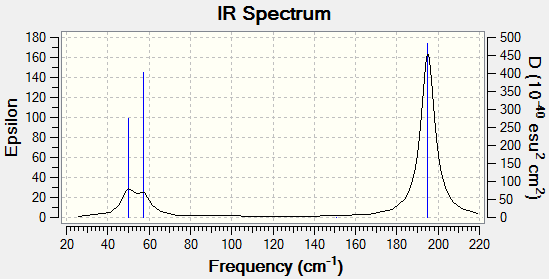
Frequency Analysis
To confirm that the energy of the molecule is at a minimum a frequency analysis can be run, using the same basis set as the geometry optimization. This is simply because the frequency analysis is the second derivative of the potential energy curve and hence if the basis set is changed, the potential energy curve changes and hence the frequency results are based on a structure with different energy, hence different geometry. The output files can be found [[2]]
The vibrational modes were calculated using the following criteria:
# freq b3lyp/lanl2dz geom=connectivity
The main vibrations are listed in Table 4.
The IR Spectrum of TlBr3 is shown below.
Apart from these main vibrations, 6 lower frequencies were also identified in the log file.
Low frequencies --- -0.0008 -0.0004 -0.0003 18.6824 19.5013 19.5013
The negative values are very close to zero, and all other frequencies are significantly lower than the first real vibrational mode of 49.98 cm-1. This confirms that the calculation has found a minimum energy and was successful.
What is a Bond?
A bond is an equilibrium point between the forces of attraction between two atoms in 3D space, and internuclear repulsion. This results in a more stable arrangement comparatively to that which free atoms experience. It has been noticed that Gaussian optimizations viewed in GaussView often result in bonds disappearing, even though a formal bond exists between the two atoms. This is a consequence of the pre-determined inter-atomic distances that GaussView uses to determine whether or not bonds should be drawn on screen. For inorganic compounds this is a frequent occurrence, because GaussView's bond databases are mostly based on organic compounds, and hence are not designed to deal with the greater internuclear distances that are frequently found to exist in inorganic compounds.
Geometric Isomers of Mo(CO)4L2
Computational modelling is often very CPU intensive and so often sacrifices have to be made to speed up the calculation time. Often one would simply reduce the basis set to improve the calculation time, however there is another route, that of simplifying the substitution about a particular atom centre. Here we will calculate the vibrational spectra of the Mo(CO)4L2 where L is PPh3, however in order to simplify the molecular complexity of PPh3, we shall substitute Cl for Ph which has a similar electronic contribution as the phenyl groups, and is also similarly sterically demanding.
Geometry Optimization of Mo(CO)4L2 Isomers
First, a rough geometry optimization was calculated using the following criteria:
# opt=loose b3lyp/lanl2mb geom=connectivity
This provided the correct geometry to the molecule, however the PCl3 needed manual editting to ensure they were in the correct conformation. For this cis conformer this was simply achieved via rearrangement of the Cl atoms such that one P-Cl bond alligned parralel to the axial bond, while the other P-Cl bond was aligned anti-parallel. For the trans isomer both PCl3 groups are eclipsed with one P-Cl bond parallel to an Mo-C bond.
These corrected conformers were then accurately optimized using the following criteria:
# opt b3lyp/lanl2dz geom=connectivity int=ultrafine scf=conver=9
The resulting output files are found [Cis Isomer] for the cis complex and [Trans Isomer] for the trans complex. The resulting configurations are given below in Table 5, along with the important bond lengths in the conformers.
| Bond | Trans Isomer | Cis Isomer | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| Bond | calculated /Å | Lit. [2] /Å | calculated /Å | literature /Å [3] | ||||||
| Mo-P | 2.44 | 2.50 | 2.51 | 2.58 | ||||||
| eq. Mo-C | 2.06 | 1.85 | 2.06 | 2.02 | ||||||
| ax. Mo-C | - | - | 2.01 | 1.97 | ||||||
| P-Cl | 2.24 | - | 2.24 | - | ||||||
| ax. C-O | - | - | 1.17 | 1.15 | ||||||
| eq. C-O | 1.17 | 1.16 | 1.18 | 1.13 | ||||||
In general the calculated bond lengths for both the cis and trans complex correlate very well with experimental values. On comparing the literature values axial and equatorial Mo-C bond lengths in the cis complex, it can be seen that the former is shorter. This is due to the trans effect, whereby the PCl33 is a good π acceptor but a poor σ donor, thus competing for back donation with other attached ligands and weakening the bond trans to it. This causes the equatorial C-O bonds to elongate comparatively to it's axial counterpart and the Mo-P and Mo-C bonds get stronger and shorter.
In the trans isomer, the PCl3 groups are in an axial position and hence are not able to exert a trans effect onto the equatorial CO bonds.
Vibrational Analysis of Mo(CO)4L2 Isomers
A frequency analysis was run on the two isomers to determine their vibrational modes. The following criteria was used:
# freq b3lyp/lanl2dz geom=connectivity int=ultrafine scf=conver=9
The results are shown in Table 6. The output files are available here: [Cis Isomer], [Trans Isomer].
| Number | Frequency (cm-1) | Literature (cm-1) | Intensity | Description | Animation | Vibrational Mode | |
|---|---|---|---|---|---|---|---|
| Trans Isomer (D4H) (Lit values [4]) | |||||||
| 1 | 4.82 | - | 0.09 | IR inactive |  |
PCl3 bend | |
| 2 | 6.11 | - | 0.00 | IR inactive | 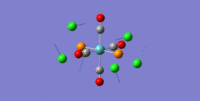 |
PCl3 bend | |
| 3 | 1950.55 | 1886 | 1475.38 | trans asymmetric C=O stretches | 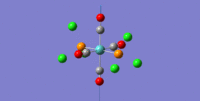 |
Eu | |
| 4 | 1951.18 | 1866 | 1466.78 | trans asymmetric C=O stretches | 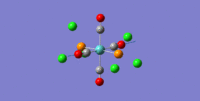 |
Eu | |
| 5 | 1966.83 | 1933 | 0.6552 | all C=O asymmetric stretches | 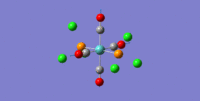 |
B1g | |
| 6 | 2031.24 | 2050 | 3.7881 | all C=O symmetric stretches | 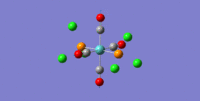 |
A1g | |
| Cis Isomer (C2V) (Lit values [5]) | |||||||
| 1 | 10.79 | - | 0.02 | IR Inactive |  |
PCl3 Scissor Bend | |
| 2 | 17.64 | - | 0.00 | IR Inactive |  |
PCl3 Twist Bend | |
| 3 | 1945.33 | 1869 | 763.47 | Asymmetric stretch of equatorial CO ligands |  |
B2 | |
| 4 | 1948.62 | 1896 | 1497.71 | Asymmetric stretch of axial CO ligands |  |
B1 | |
| 5 | 1958.36 | 1924 | 632.14 | Symmetric stretch of equatorial carbonyls |  |
A1 | |
| 6 | 2023.28 | 2026 | 598.27 | Symmetric stretch of axial carbonyls |  |
A1 | |
Table 7 shows the calculated IR spectra of both isomers:
| Trans Isomer | Cis Isomer |
|---|---|
 |

|
The number of C=O vibrational bands is related to the symmetry of the complex, so one would expect more peaks to appear on an IR spectra of the less symmetric cis isomer, with C2V symmetry, than of the trans isomer which has D4H symmetry. In this case we expect 4 carbonyl stretching frequencies for the cis complex, and one from the trans complex. There are in fact 4 carbonyl stretching frequencies for both complexes, however only two are of high intensity for the trans complex and they are effectively degenerate hence appearing as one band.
For the cis complex all four frequencies are non degenerate and compare to the literature values well. The vibrational frequency of uncoordinated C=O is 2143cm-1[6]. The coordinated carbonyls all have lower stretching frequencies due to Mo-C back bonding, whereby the C=O π* orbital receives electron density from the metal d-orbitals and hence weakens the bond, lowering it's vibrational frequency. The carbonyls in the trans isomer are all in the same environment and so experience the same amount of back bonding, however the carbonyls in the cis complex are in different environments and hence back bonding varies.
Exploring Bonding in Main Group Halides
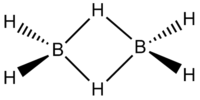
Group 13 Halides such as BH3 can act as Lewis Acids and as such they are regularly found to dimerize through the use of an electron deficient 3 centre - 2 electron bond. The most famous example of this is diborane, B2H6, whose structure could not be discerned by VSEPR theory and was only rationalised through consideration of Molecular Orbital Theory.
The dimers have two environments within the molecule, the bridging and terminal positions. It is well known that halogens preferentially occupy the bridging positions
over hydrocarbon or hydride groups[7], however this mini project aims to discern what happens when two different halogens are present in the dimer.
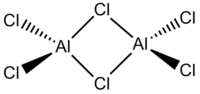
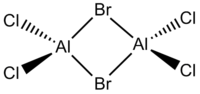
In this project the following dimeric group 13 halides will be analysed.
| Diborane | Dialuminium chloride | (Al2Cl4(μ2-Br) | cis-(Al2Cl2Br2(μ2-Cl)2 | trans-Al2Cl2Br2(μ2-Cl)2 |
|---|---|---|---|---|
 |
 |
 |
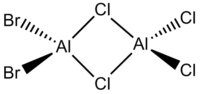 |
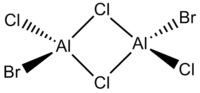
|
Structural Properties
Diborane was drawn in gaussview and it's geometry was optimized using the following criteria:
# opt b3lyp/lanl2dz geom=connectivity int=ultrafine scf=conver=9
The output files are available here: [(B2H6)], [chloride (Al2Cl6)], [2Cl4(μ2-Br)2], [cis-Al2Cl2Br2(μ2-Cl)2], [trans-Al2Cl2Br2(μ2-Cl)2</sub]. Table 9 shows the output of the calculations.
| Structure |
|
|
|
|
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | Diborane (B2H6) | Dialuminium chloride (Al2Cl6) | Al2Cl4(μ2-Br)2 | cis-Al2Cl2Br2(μ2-Cl)2 | trans-Al2Cl2Br2(μ2-Cl)2 | |||||||||||||||
| Bond Lengths /Å | B-(μ2-H): 1.34; B-H: 1.19 | Al-(μ2-Cl): 2.42; Al-Cl: 2.18 | Al-(μ2-Br): 2.59; Al-Cl: 2.18 | Al-(μ2-Cl): 2.41; Al-Cl: 2.18; Al-Br: 2.34 | Al-(μ2-Cl): 2.42; Al-Cl: 2.18; Al-Br: 2.35 | |||||||||||||||
| Literature / Å [8] | - | - | Al-(μ2-Br): 2.33 | Al-Br(t): 2.21; Al-Cl(t): 2.06; Al-(μ2-Cl): 2.21 | - | |||||||||||||||
| Energy | -53.2595 (-139,832.7970 kJ / mol) | -94.0203 (-246,850.2619 kJ / mol) | -90.4336 (-237,433.2824 kJ / mol) | -90.4452 (-237,463.8382 kJ / mol) | -90.4449 (-237,463.0505 kJ / mol) |
Firstly it can be seen that the relative order of stability between the isomers is:
- cis-(Al2Cl2Br2(μ2-Cl)2 is most stable.
- trans-Al2Cl2Br2(μ2-Cl)2 is in the middle.
- (Al2Cl4(μ2-Br)2 is least stable.
Intuitively one would expect that with the inclusion of the the larger bromine atoms, the structure would prefer to adopt the trans configuration thus maximising the large bromine atom separation and reducing steric repulsion. Or perhaps one might expect that the A-(μ2-Br) might be preferred, which is a better π-donor into the lewis acidic aluminium atom. Obviously neither of these two situations are the case, and further analysis is needed to rationalise the evident behaviour.
The Al-Cl (terminal) bond length of 2.18Å seems to be independent upon the position of the bromine atom in the molecule, or whether bromine is in the molecule at all as shown by the Al2Cl6 dimer. This suggests that chlorine is sufficiently small to be unaffected by internuclear repulsion from the Br atom. Comparatively upon the isomerism from cis to trans Al2Cl4Br4, the Al-Br bond distance changes by 0.01Å. Although is change is small, a similar observation can be made when the briding atoms are changed from Cl to Br. The change in bond length on moving from Al-Cl(terminal) to Al-(μ2-Cl) is 0.23-0.24Å. Comparatively on moving from Al-Br (terminal) to Al- (μ2-Br)2 the change in bond length is 0.25-0.26A, suggesting slightly larger steric crowding and hence a weakening of the Al-Br bonds. As orbital overlap is distance dependant, this increase in bond distance will also result in a less stable system. On the other hand, due to it's smaller size chlorine is better able to accommodate this change in distance upon bridging, and hence is less affected by the decrease in orbital overlap thus providing a rational over why bromine bridged systems are so unstable comparatively to chlorine bridged systems.
Vibrational Properties
The vibrational modes of cis-(Al2Cl2Br2(μ2-Cl)2, trans-Al2Cl2Br2(μ2-Cl)2 and Al2Cl4(μ2-Br)2 were calculated to analyse the effects of halogen substitution on vibrational energy.
The followingh critera was used to calculate the vibrational properties of each molecule:
# freq b3lyp/lanl2dz geom=connectivity int=ultrafine scf=conver=9
The results are available here: [cis-(Al2Cl2Br2(μ2-Cl)2], [trans-Al2Cl2Br2(μ2-Cl)2], [|Al2Cl4(μ2-Br)2].
Only modes involving the effective movement of the aluminium centres will be analysed. Table 10 present the calculated results.
Both modes 18 and 15 both result in the Al2Cl4(μ2-Br)2 vibration being 30-40 cm-1 higher than the trans-Al2Cl2Br2(μ2-Cl)2 vibration. The opposite is true for modes 12 and 14, which are both 30-40cm-1 lower for the Al2Cl4(μ2-Br)2 vibration than the trans-Al2Cl2Br2(μ2-Cl)2 vibration. This is unexpected, since one would expect that an increase of reduced mass about the central bridging atoms would decrease the frequency of vibration.
There are 2 more active vibrational modes present in the cis isomer and this is likely due to the reduction in symmetry on moving from the bridged or trans systems. Mode 18 matches the same Mode in both the bridged and trans isomer which indicates that this vibrational mode is independent of the non-bridging halogen atoms.
Table 12 shows the vibrational spectrum for each isomer.
| (Al2Cl4(μ2-Br)2 | cis-(Al2Cl2Br2(μ2-Cl)2 | trans-Al2Cl2Br2(μ2-Cl)2 |
|---|---|---|
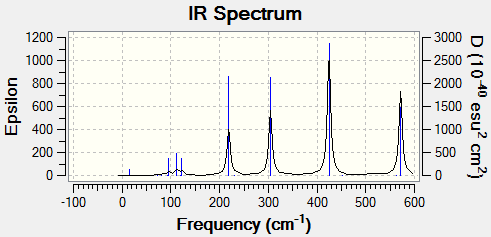 |
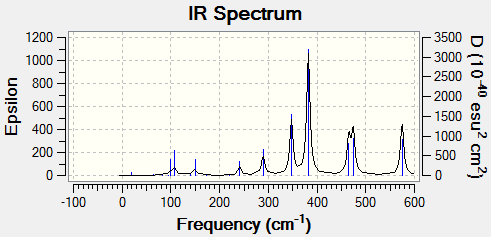 |
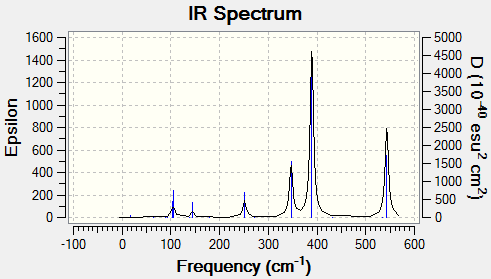
|
Charge Distribution of Bridging Compounds
The Charge distribution for each isomer was calculated in parallel to the MO calculation in the next section. Table 13 presents these results.
| (Al2Cl4(μ2-Br)2 | cis-(Al2Cl2Br2(μ2-Cl)2 | trans-Al2Cl2Br2(μ2-Cl)2 |
|---|---|---|
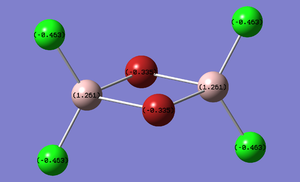 |
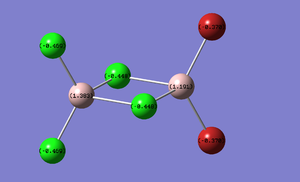 |
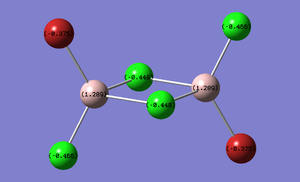
|
As expected the charge distribution mirrors that of the Pauling electronegativity of each atom. This diagram shows that the Aluminium atom is electron deficient comparatively to the electronegative chlorine and bromine atoms. As expected Bromine has a greater percentage of the charge distribution than chlorine.
Molecular Orbitals of Bridging Compounds
The Molecular Orbitals were calculated using the following criteria.
# rb3lyp/lanl2dz pop=(nbo,full) geom=connectivity
The output files are availible here: [Molecule 1], [Molecule2], [Molecule 3], [Molecule 4], [Molecule 5] . The results are presented in Table 14.
Bromine is larger and hence more diffuse than chlorine. Bonds adjoined to bromine are longer, and hence orbital coupling is less strong. This trend can be seen in a number of MO's, but is particularly evident in MO 1, where the S like overlap between the two bridging atoms is more effective for the chlorine isomers than for bromine isomers.
Stabilization energy also depends on the difference in energy between the two orbitals. A smaller difference in energy creates a greater stabilisation. A molecule will be more stable if two atoms of the same type have overlapping orbitals, compared to two atoms of different type. If two halogens of the same type are situated terminal to each other, then the resulting orbital stabilisation will be greater than if the two atoms were different.
Mo's 3-6 show that the cis configuration has better overlap between the terminal substituents. This behaviour is also shown in MO's 17-24. Clearly, the cis isomer has two identical atoms adjacent to each other on the terminal positions, which leads to a more energetically favourable overlap than that found in the trans isomer, resulting in the cis isomer having a lower overall energy than the trans isomer.
Conclusion
It was found that the cis isomer of Al2Cl4Br2 was more stable than both the trans and the Br-bridging isomer. Measurement of bond lengths indicated that steric hindrance would likely disfavour the bridging isomer, however neither analysis of the vibrational frequencies or charge distribution ave any insight on why the cis-isomer was thermodynamically favoured. Visualizing and comparing the calculated MO's allowed the rationalisation of the relative stability of the dimers.
References
- ↑ J. Glaser, G. Johansson. Acta Chemica Scandinavica A 36 (1982) 125-135[1]
- ↑ Hogarth, G.; Norman, T.; Inorganica Chimica Acta. 1997, 254, 167-171.
- ↑ Cotton, F. A.; Darensbourg, D. J.; Klein, S.; Kolthammer, B. W. S.; Inorg. Chem., 1982, 21, 294-299.
- ↑ Darensbourg, D.; Inorg. Chem., 1979, 18, 14-17.
- ↑ Ardon, M.; J. Chem. Educ., 2002, 79, 1249-1251.
- ↑ http://chem-faculty.lsu.edu/stanley/webpub/4571-chap3-carbonyls.pdf
- ↑ P. Lickiss, 2nd Year Main Group Chemistry, 2011, pp.14
- ↑ K. Wade, J. Chem. Educ., 49, 1972, pp. 502-505